It was an absolutely wonderful trip. The motions were dynamic and thoughtful, required extensive amounts of knowledge about the kinds of principles that governments are thinking of upholding, which is why one of the staple arguments in any debate was why the government is in a position to make any sort of change at all: does it have the right to prosecute communities for complicity in honour killings for example.
One of my favourite motions which I got to adjudicate was
THW grant those diagnosed with terminal illnesses the right to access treatments that have not completed clinical testing.
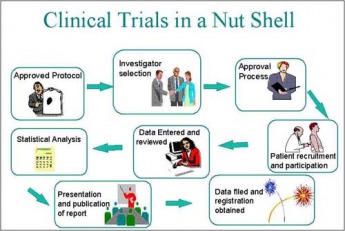
Sure, the issues inherent to this debate are not about the nature of clinical trials really, but a case could be made (and was made in the debate I sat through) that by granting such a right to cancer patients for instance, the process upon which clinical trials relies could not be carried out satisfactorily. But essentially, this is a debate about whether people who really need a life-saving drug should be allowed to take the massive risk that is an untested drug or not. Plenty of horror stories are relevant here, such as that story about Thalidomide which was tested on pregnant mothers and resulted in deformed children - okay, lets ignore the fact that pregnancy is hardly a terminal illness - the point is that clinical trials often have heinous unforeseen circumstances. But how else do you put a safe drug on the market?
Often we are told that there are alternatives: you can try them on animals for instance. Clearly, there is no really ethical way to test drugs and come up with a valid assessment of how the drug works, its side effects etc.
I will be writing an essay about clinical trials and putting it up in the Work Section over the holidays. If you want to know more about clinical trials, what common methodologies they rely on, where they usually get the money to carry through from etc. here's the Wikipedia entry on clinical trials.
Kamil
 RSS Feed
RSS Feed